Abstract
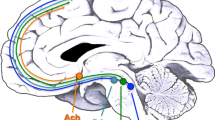
Using multimodal neuroimaging in humans, we demonstrate specific interactions between prefrontal activity and midbrain dopaminergic synthesis. A common V(108/158)M substitution in the gene for catecholamine-O-methyltransferase (COMT), an important enzyme regulating prefrontal dopamine turnover, predicted reduced dopamine synthesis in midbrain and qualitatively affected the interaction with prefrontal cortex. These data implicate a dopaminergic tuning mechanism in prefrontal cortex and suggest a systems-level mechanism for cognitive and neuropsychiatric associations with COMT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Sesack, S.R., Carr, D.B., Omelchenko, N. & Pinto, A. Ann. NY Acad. Sci. 1003, 36–52 (2003).
Seamans, J.K. & Yang, C.R. Prog. Neurobiol. 74, 1–58 (2004).
Grace, A.A. J. Neural Transm. Gen. Sect. 91, 111–134 (1993).
Williams, G.V. & Goldman-Rakic, P.S. Nature 376, 572–575 (1995).
Weinberger, D.R. Arch. Gen. Psychiatry 44, 660–669 (1987).
Chen, J. et al. Am. J. Hum. Genet. 75, 807–821 (2004).
Gogos, J.A. et al. Proc. Natl. Acad. Sci. USA 95, 9991–9996 (1998).
Egan, M.F. et al. Proc. Natl. Acad. Sci. USA 98, 6917–6922 (2001).
Mattay, V.S. et al. Proc. Natl. Acad. Sci. USA 100, 6186–6191 (2003).
Akil, M. et al. J. Neurosci. 23, 2008–2013 (2003).
Meyer-Lindenberg, A. et al. Nat. Neurosci. 5, 267–271 (2002).
Meyer-Lindenberg, A. et al. Am. J. Psychiatry 158, 1809–1817 (2001).
Gjedde, A. et al. Proc. Natl. Acad. Sci. USA 88, 2721–2725 (1991).
Matsumoto, M. et al. Neuroscience 116, 127–137 (2003).
Meador-Woodruff, J.H., Damask, S.P. & Watson, S.J., Jr. Proc. Natl. Acad. Sci. USA 91, 8297–8301 (1994).
Acknowledgements
We thank D. Sarpal, P. Koch and A. Bonner-Jackson for research assistance, M. Akil and V. Mattay for helpful discussion of this manuscript vis à vis their own results, A. Grace for helpful comments on the interpretation of the data, T. Goldberg for neuropsychological testing and R. Carson for expertise in PET kinetic modeling.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Relationship between dopaminergic stimulation and prefrontal cortex activity, drawn schematically after Goldman-Rakic et al. and Mattay et al. In the latter study, the differential effect of an acute increase in dopaminergic tone induced by amphetamine was used to probe the position on the u-shaped curve in the setting of acute pharmacological modulation, which led to improved function and prefrontal efficiency in val-allele carriers, but deteriorating function in met-homozygotes. (JPG 48 kb)
Supplementary Fig. 2
Task-related activations and deactivations. Effect of task - significant activations (red) and deactivations (blue), comparing the working memory (2-back) condition with its sensorimotor control (0-back). Highlighted voxels are significant at theP⩽0.05 corrected level (see Supplementary Tables 1 and 2). (JPG 99 kb)
Supplementary Fig. 3
Hypothetical fit of data to “inverted-u” response curve. “Inverted-u” shaped relationship between observed left DLPFC 0-back rCBF and dopamine synthesis rate, hypothetically assuming that a given rate of midbrain dopamine neuronal activity and dopamine synthesis (Ki) will result in twice as much prefrontal dopamine in met-homozygotes (see discussion in Chen et al. 2004); to reflect this, Kis were doubled for met-homozygotes. Second-order polynomial fit curve shown. Datapoints for val-carriers shown as filled circles, met-homozygotes as empty circles. (JPG 33 kb)
Supplementary Table 1
Further subject information and ROI measurements, by genotype. (PDF 48 kb)
Supplementary Table 2
Working-memory task related activations. (PDF 42 kb)
Supplementary Table 3
Working-memory task related deactivations. (PDF 42 kb)
Supplementary Table 4
Correlations of PFC rCBF with midbrain F-DOPA Ki. (PDF 45 kb)
Rights and permissions
About this article
Cite this article
Meyer-Lindenberg, A., Kohn, P., Kolachana, B. et al. Midbrain dopamine and prefrontal function in humans: interaction and modulation by COMT genotype. Nat Neurosci 8, 594–596 (2005). https://doi.org/10.1038/nn1438
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1438
This article is cited by
-
Behavioural and functional evidence revealing the role of RBFOX1 variation in multiple psychiatric disorders and traits
Molecular Psychiatry (2022)
-
The changing opioid crisis: development, challenges and opportunities
Molecular Psychiatry (2021)
-
Distinct roles for dopamine clearance mechanisms in regulating behavioral flexibility
Molecular Psychiatry (2021)
-
Brain network dynamics during working memory are modulated by dopamine and diminished in schizophrenia
Nature Communications (2021)
-
Dopamine promotes aggression in mice via ventral tegmental area to lateral septum projections
Nature Communications (2021)